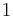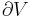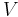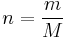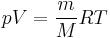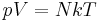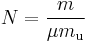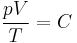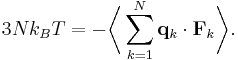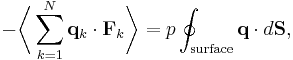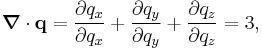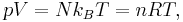Ideal gas law
| Thermodynamics | |||||||||||||||||||||||||||||||||||||||||||||||

|
|||||||||||||||||||||||||||||||||||||||||||||||

The ideal gas law is the equation of state of a hypothetical ideal gas. It is a good approximation to the behavior of many gases under many conditions, although it has several limitations. It was first stated by Émile Clapeyron in 1834 as a combination of Boyle's law and Charles's law.[1] It can also be derived from kinetic theory, as was achieved (apparently independently) by August Krönig in 1856[2] and Rudolf Clausius in 1857.[3]
The state of an amount of gas is determined by its pressure, volume, and temperature. The modern form of the equation is:
where p is the absolute pressure of the gas; V is the volume; n is the amount of substance; R is the gas constant; and T is the absolute temperature.
In SI units, p is measured in pascals; V in cubic metres; n in moles; and T in kelvin. R has the value 8.314472 J·K−1·mol−1 in SI units[4]).
The temperature given in the equation of state must be an absolute temperature that begins at absolute zero. In the metric system of units, we must specify the temperature in Kelvin (not degrees Celsius). In the Imperial system, absolute temperature is in Rankine (not degrees Fahrenheit). [5]
Contents |
Deviations from real gases
The equation of state given here applies only to an ideal gas, or a real gas that behaves like an ideal gas. There are in fact many different forms for the equation of state for different gases. Since it neglects both molecular size and intermolecular attractions, the ideal gas law is most accurate for monatomic gases at high temperatures and low pressures. The neglect of molecular size becomes less important for lower densities, i.e. for larger volumes at lower pressures, because the average distance between adjacent molecules becomes much larger than the molecular size. The relative importance of intermolecular attractions diminishes with increasing thermal kinetic energy i.e., with increasing temperatures. More detailed equations of state, such as the van der Waals equation, allow deviations from ideality caused by molecular size and intermolecular forces to be taken into account.
A residual property is defined as the difference between a real gas property and an ideal gas property, both considered at the same pressure, temperature, and composition.
Alternative forms
Molar form
As the amount of substance could be given in mass instead of moles, sometimes an alternative form of the ideal gas law is useful. The number of moles (n) is equal to the mass (m) divided by the molar mass (M):
By replacing 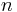 , we get:
, we get:
from where
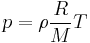 .
.
This form of the ideal gas law is very useful because it links pressure, density ρ = m/V, and temperature in a unique formula independent from the quantity of the considered gas.
Engineering
Engineers use a slightly different form of the equation of state that is specialized for a particular gas and written in terms of specific volume.
A variation of the gas constant R is used, which is equal to the universal gas constant divided by the mass per mole of the gas. The value of the new constant depends on the type of gas, as opposed to the universal gas constant which is the same for all gases. In most engineering fields,  is written with a bar, as
is written with a bar, as 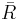 , so that a new symbol
, so that a new symbol  is defined by
is defined by 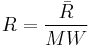 , where
, where 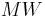 is the molecular weight.
is the molecular weight.
If we divide both sides of the general ideal gas equation by the mass of the gas, the volume becomes the specific volume, which is the inverse of the gas density. The equation of state can be written in terms of the specific volume or in terms of the air density as
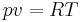 , or
, or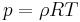 ,
,
where
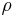 is the density [kg/m3] and
is the density [kg/m3] and 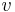 is the specific volume [m3/kg].
is the specific volume [m3/kg].
Statistical mechanics
In statistical mechanics the following molecular equation is derived from first principles:
Here k is the Boltzmann constant, and N is the actual number of molecules, in contrast to the other formulation, which uses n, the number of moles. This relation implies that Nk = nR, and the consistency of this result with experiment is a good check on the principles of statistical mechanics.
From here we can notice that for an average particle mass of μ times the atomic mass constant mu (i.e., the mass is μ u)
and since ρ = m/V, we find that the ideal gas law can be rewritten as:
Applications to thermodynamics processes
The table below essentially simplifies the ideal gas equation for a particular processes, thus making this equation easier to solve using numerical methods.
A thermodynamic process is defined as a system that moves from state 1 to state 2, where the state number is denoted by subscript. As shown in the first column of the table, basic thermodynamic processes are defined such that one of the gas properties (p, V, T, or S) is constant throughout the process.
For a given thermodynamics process, in order to specify the extent of a particular process, one of the properties ratios (listed under the column labeled "known ratio") must be specified (either directly or indirectly). Also, the property for which the ratio is known must be distinct from the property held constant in the previous column (otherwise the ratio would be unity, and not enough information would be available to simplify the gas law equation).
In the final three columns, the properties (p, V, or T) at state 2 can be calculated from the properties at state 1 using the equations listed.
| process | Constant | Known ratio | p2 | V2 | T2 |
|---|---|---|---|---|---|
| Isobaric process |
|
|
p2 = p1 | V2 = V1(V2/V1) | T2 = T1(V2/V1) |
|
|
p2 = p1 | V2 = V1(T2/T1) | T2 = T1(T2/T1) | ||
| Isochoric process |
|
|
p2 = p1(p2/p1) | V2 = V1 | T2 = T1(p2/p1) |
|
|
p2 = p1(T2/T1) | V2 = V1 | T2 = T1(T2/T1) | ||
| Isothermal process |
|
|
p2 = p1(p2/p1) | V2 = V1/(p2/p1) | T2 = T1 |
|
|
p2 = p1/(V2/V1) | V2 = V1(V2/V1) | T2 = T1 | ||
| Isentropic process (Reversible adiabatic process) |
|
|
p2 = p1(p2/p1) | V2 = V1(p2/p1)−1/γ | T2 = T1(p2/p1)(γ − 1)/γ |
|
|
p2 = p1(V2/V1)−γ | V2 = V1(V2/V1) | T2 = T1(V2/V1)(1 − γ) | ||
|
|
p2 = p1(T2/T1)γ/(γ − 1) | V2 = V1(T2/T1)1/(1 − γ) | T2 = T1(T2/T1) |
^ a. In an isentropic process, system entropy (S) is constant. Under these conditions, p1V1γ = p2V2γ, where γ is defined as the heat capacity ratio, which is constant for an ideal gas. The value used for γ is typically 1.4 for diatomic gases like nitrogen (N2) and oxygen (O2), (and air, which is 99% diatomic). Also γ is typically 1.6 for monatomic gases like the noble gases helium (He), and argon (Ar). In internal combustion engines γ varies between 1.35 and 1.15, depending on constitution gases and temperature.
Derivations
Empirical
The ideal gas law can be derived from combining two empirical gas laws: the combined gas law and Avogadro's law. The combined gas law states that
where C is a constant which is directly proportional to the amount of gas, n (Avogadro's law). The proportionality factor is the universal gas constant, R, i.e. C = nR.
Hence the ideal gas law
Theoretical
The ideal gas law can also be derived from first principles using the kinetic theory of gases, in which several simplifying assumptions are made, chief among which are that the molecules, or atoms, of the gas are point masses, possessing mass but no significant volume, and undergo only elastic collisions with each other and the sides of the container in which both linear momentum and kinetic energy are conserved.
From statistical mechanics
Let q = (qx, qy, qz) and p = (px, py, pz) denote the position vector and momentum vector of a particle of an ideal gas, respectively. Let F denote the net force on that particle. Then the time average momentum of the particle is:
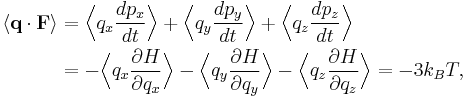
where the first equality is Newton's second law, and the second line uses Hamilton's equations and the equipartition theorem. Summing over a system of N particles yields
By Newton's third law and the ideal gas assumption, the net force on the system is the force applied by the walls of their container, and this force is given by the pressure p of the gas. Hence
where dS is the infinitesimal area element along the walls of the container. Since the divergence of the position vector q is
the divergence theorem implies that
where dV is an infinitesimal volume within the container and V is the total volume of the container.
Putting these equalities together yields
which immediately implies the ideal gas law for N particles:
where n = N/NA is the number of moles of gas and R = NAkB is the gas constant.
The readers are referred to the comprehensive article Configuration integral (statistical mechanics) where an alternative statistical mechanics derivation of the ideal-gas law, using the relationship between the Helmholtz free energy and the partition function, but without using the equipartition theorem, is provided.
See also
- Combined gas law
- Van der Waals equation
- Boltzmann's constant
- Configuration integral
References
- ↑ Clapeyron, E. (1834). "Mémoire sur la puissance motrice de la chaleur". Journal de l'École Polytechnique XIV: 153–90. (French) Facsimile at the Bibliothèque nationale de France (pp. 153–90).
- ↑ Krönig, A. (1856). "Grundzüge einer Theorie der Gase". Annalen der Physik 99: 315–22. doi:10.1002/andp.18561751008. (German) Facsimile at the Bibliothèque nationale de France (pp. 315–22).
- ↑ Clausius, R. (1857). "Ueber die Art der Bewegung, welche wir Wärme nennen". Annalen der Physik und Chemie 100: 353–79. doi:10.1002/andp.18571760302. (German) Facsimile at the Bibliothèque nationale de France (pp. 353–79).
- ↑ .R is also sometimes expressed as 0.08205746 L·Atm·K−1·mol−1
- ↑ "Equation of State". http://www.grc.nasa.gov/WWW/K-12/airplane/eqstat.html.
Further reading
- Davis and Masten Principles of Environmental Engineering and Science, McGraw-Hill Companies, Inc. New York (2002) ISBN 0-07-235053-9
- Website giving credit to Benoît Paul Émile Clapeyron, (1799-1864) in 1834
|
||||||||||||||||||||||||||